

Specialty pharmacy plays a critical role in caring for patients with complex, chronic conditions – and employers recognize its value. 98% of employers say specialty pharmacy…

Beginning in late July 2026, significant new tariffs are expected on imported patented drugs and key ingredients under Section 232 of the Trade Expansion Act. These tariffs – up to 100% for some manufacturers – are intended to…

Pharmacy Benefit Managers (PBMs) are often discussed in terms of cost – but their value extends far beyond savings. On average, PBMs help patients and employers save over $1,100 per person each year by negotiating drug prices…
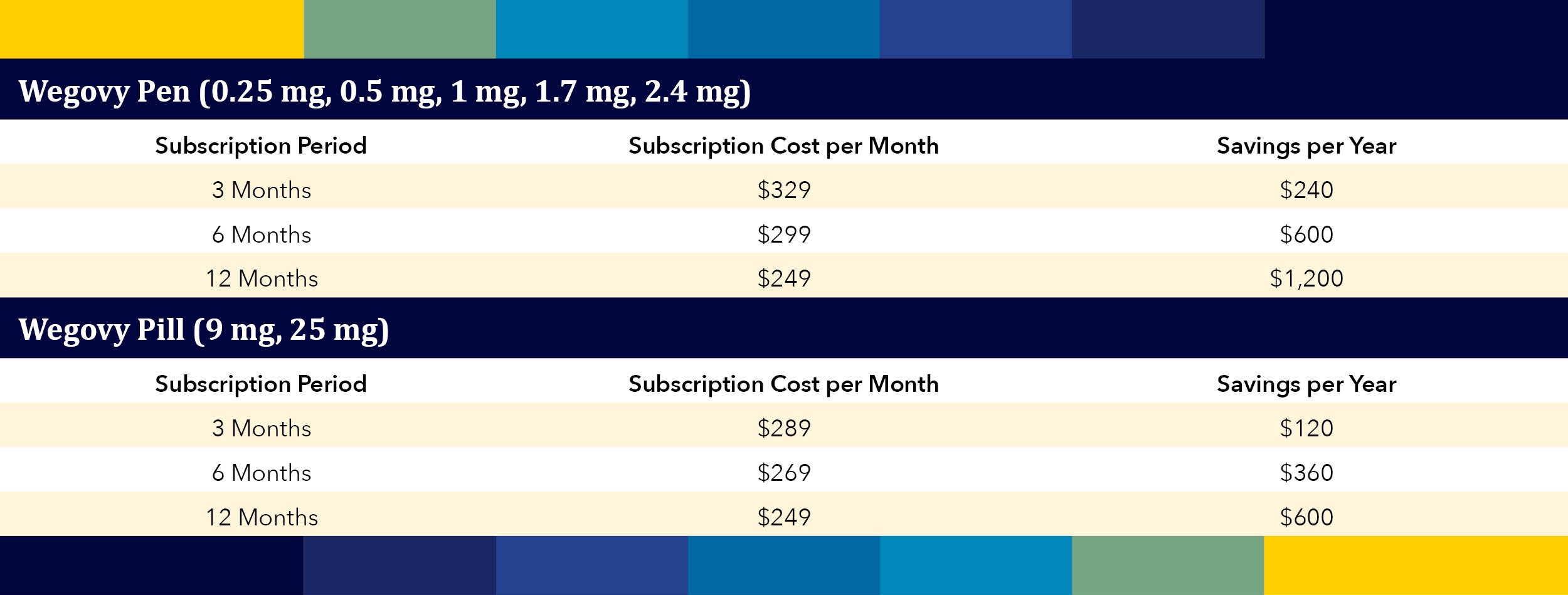
On April 1, 2026, the FDA approved Eli Lilly’s Foundayo, the second oral GLP‑1 for weight loss – offering fewer administration restrictions and achieving one of the fastest new‑drug approvals in decades.…

On March 26, 2026, the FDA approved Awiqli® (insulin icodec abae) from Novo Nordisk – the first once‑weekly basal insulin for adults with type 2 diabetes. This milestone represents a meaningful shift in…





