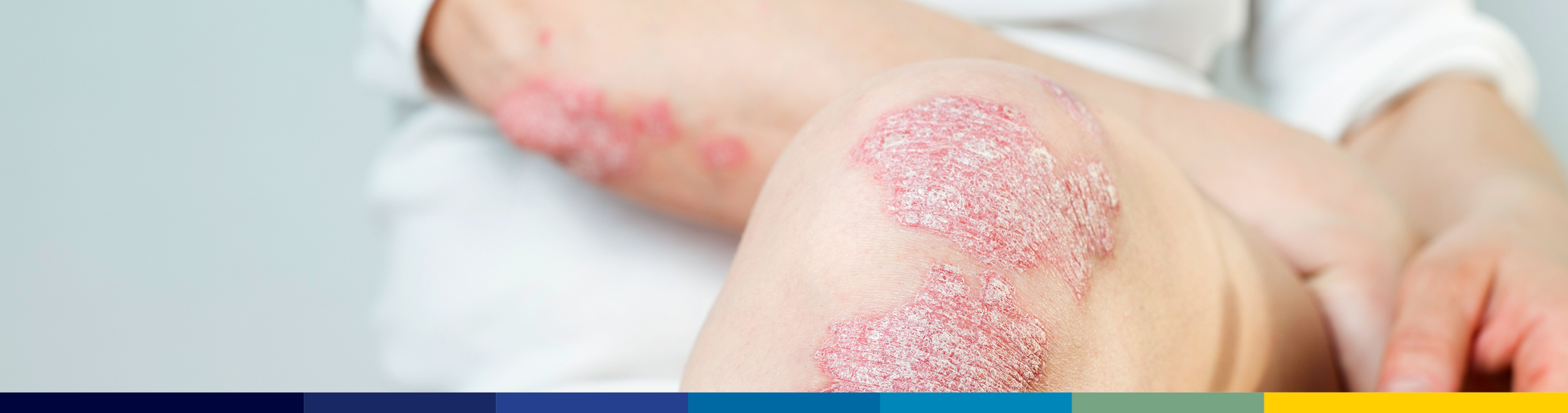
On February 23, 2024, the FDA approved Alvotech and Teva’s Simlandi (adalimumab-ryvk; formerly AVT02) injection, as an interchangeable biosimilar to high-concentration, citrate-free Humira. The high-concentration formulation accounts for over 85% of Humira’s market share. Simlandi can be substituted for Humira at the dispensing pharmacy, subject to state laws. Pricing has not yet been announced but the drug manufacturer plans to launch very soon, however a specific date has not yet been given. ProAct completes individual reviews of new biosimilar products for potential formulary placement and considers many factors when evaluating new biosimilars including formulations/concentrations, pricing, and the ability to supply the market and drive conversions.
https://www.centerforbiosimilars.com/view/fda-approves-tenth-adalimumab-biosimilar-simlandi